Original Article – DOI: 10.33594/000000861
Neurosignals (33): 1 - 18
Accepted: 02.04.2026 - Pulished online: 17.04.2026
A Comprehensive Review of Melatonin as Multi-Pathway Neuroprotectant Against Methamphetamine-Induced Programmed Cell Death: Discovery of the Circadian-Ferroptosis Axis
bDepartment of Physiology, Faculty of Science, Mahidol University, Bangkok, Thailand, 272 Rama VI Road, Ratchathewi District, Bangkok 10400, Thailand,
cPrincess Agrarajakumari Faculty of Nursing, Chulabhorn Royal Academy, 906 Thung Song Hong, Lak Si, Bangkok 10210, Thailand,
dResearch Center for Integrative Biomedical Science in Medicine and Health, Chulabhorn Royal Academy, 906 Thung Song Hong, Lak Si, Bangkok 10210, Thailand
Keywords
Abstract
Introduction: Methamphetamine (METH) abuse affects 34 million individuals globally, causing severe neurotoxicity through multiple programmed cell death (PCD) pathways. No approved pharmacotherapies exist. We comprehensively examined melatonin's neuroprotective mechanisms against METH-induced apoptosis, pyroptosis, necroptosis, and ferroptosis. Methods: A comprehensive review of preclinical studies examining METH neurotoxicity mechanisms and melatonin's protective effects across all PCD pathways. This study summarized molecular targets, signaling cascades, and their translational potential. Results: METH activates four distinct PCD pathways: (1) apoptosis via mitochondrial dysfunction and ER stress (increased CHOP, caspase-12 and cytochrome c release); (2) pyroptosis via NLRP3/NLRP1 inflammasome assembly (increased caspase-1 and IL-1β/IL-18); (3) necroptosis via RIPK1/RIPK3/MLKL signaling; and (4) ferroptosis via GPX4 downregulation, iron dysregulation, and lipid peroxidation. Melatonin simultaneously inhibits all pathways through antioxidant activity, mitochondrial protection, anti-inflammatory effects, and direct pathway targeting. Critically, we identified the concept of circadian-ferroptosis axis: METH disrupts circadian rhythms, enhancing ferroptosis through clockophagy (selective autophagy-mediated ARNTL/BMAL1 degradation). Melatonin provides dual protection through circadian restoration and direct ferroptosis inhibition. Studies demonstrate ARNTL-dependent anti-ferroptotic effects, as ARNTL knockdown abolishes melatonin's protection, revealing mechanistic integration between chronobiotic and neuroprotective actions. Conclusions: Melatonin exhibits exceptional multi-target neuroprotection against METH toxicity. The circadian-ferroptosis-METH-melatonin nexus represents an emerging conceptual framework with implications for neurodegenerative diseases and substance use disorders. Clinical trials are urgently warranted given excellent safety profile and critical unmet need.
Emerging Concept
Accumulating evidence reveals a previously underappreciated link between circadian rhythm disruption and ferroptosis-mediated cell death. Melatonin, through its regulation of core clock genes including ARNTL/BMAL1, REV-ERB, and Period, modulates ferroptosis susceptibility in a time-dependent manner. This circadian-ferroptosis axis may represent a promising therapeutic target for METH–induced neurotoxicity.
Introduction
Programmed cell death (PCD) is a genetically natural mechanism that plays a crucial role in the development of multicellular organism in order to allow normal formation of the organ systems including mammalian neural structures and in tissue homeostasis. However, PCD can also be observed in the pathogenesis of various neurological diseases where PCD signaling cascades are abnormally regulated [1-3]. Methamphetamine (METH) is the most common psychostimulant with highly addictive properties. Because of its wide use and associated health concerns, its neurotoxic effects are the focus of several research efforts to develop effective treatments. METH is a strong stimulant that can impact behaviors and cognitive functions of abusers, such as sleep disorders and neurodegeneration [4, 5]. The neurotoxic effects of METH are mediated via several mechanisms, including the direct damage to various brain areas, mitochondrial dysfunction, increased oxidative and endoplasmic reticulum (ER) stress, induced neuronal excitability, and neuroinflammation, resulting in PCD. The molecular mechanisms responsible for brain damage caused by METH involve many different processes, and there is now extensive evidence that melatonin can reverse these harmful effects [3]. In this review, we summarize the deleterious effects of METH on PCD processes, and the beneficial role of melatonin in counteracting those effects.
Method
A comprehensive literature search was conducted in MEDLINE/PubMed, Scopus, and Google Scholar for studies published between 2021 and 2026, using the keywords ‘methamphetamine,’ ‘melatonin,’ ‘programmed cell death,’ ‘neurodegeneration,’ ‘apoptosis,’ ‘pyroptosis,’ ‘necroptosis,’ ‘ferroptosis,’ ‘panoptosis,’ and ‘circadian rhythm. Studies were included if they examined the neuroprotective effects of melatonin against METH-induced neurotoxicity or cell death, particularly those providing mechanistic insights into pathways such as circadian regulation, oxidative stress, mitochondrial dysfunction, apoptosis, and ferroptosis. Eligible studies were limited to original and peer-reviewed experimental research (in vitro, in vivo, or ex vivo) published in English. Studies were excluded if they did not involve METH-induced toxicity or melatonin intervention, lacked mechanistic data, focused on non-neuronal systems without clear neuroprotective relevance, or were non-original articles (e.g., reviews, editorials, or conference abstracts) or non-English publications.
Methamphetamine (METH)-Induced Neuronal Damage
Chronic use of METH has been associated with numerous adverse effects on the brain, including neurotoxicity, neuroinflammation, and cognitive dysfunction [6-8] (Fig. 1). Neurotoxicity refers to the damage caused to brain cells, which can lead to impairment in various cognitive and behavioral functions. The impact of METH-induced neurotoxicity is particularly evident in several regions such as the striatum and prefrontal cortex, causing degeneration of dopamine-producing neurons in the striatum [9]. These can result in impaired motor function, altered reward processing, and potential addiction-related behaviors. METH-induced neurotoxicity in the prefrontal cortex can disrupt the balance of neurotransmitters and impair the communication between neurons. Elucidation of the mechanisms underlying METH-induced neurotoxicity is imperative for the development of effective therapeutic strategies for METH addiction, which is strongly associated with neurological dysfunction. Growing evidence indicates that METH neurotoxicity is mediated through several mechanisms, including oxidative stress, mitochondrial dysfunction, excitotoxicity, neuroinflammation, and activation of PCD [10]. METH is a potent releaser of monoamines, including dopamine, serotonin, and norepinephrine, resulting in a surge in the levels of these neurotransmitters. Prolonged exposure to METH reduces dopamine levels and its associated metabolites and induces persistent damage to dopaminergic and serotonergic axon terminals in key brain regions, including the striatum, hippocampus, and prefrontal cortex. Besides monoaminergic disruption, METH neurotoxicity also affects other neurotransmitter systems, particularly serotonin and glutamate [11-15]. These widespread neurochemical alterations contribute to the complex symptom profile of METH toxicity and highlight the profound and long-lasting impact of this substance on both the structural and functional integrity of the brain. Moreover, METH neurotoxicity promotes the production of reactive oxygen species (ROS), reduces antioxidant capacity, and impairs mitochondrial function, thereby initiating and exacerbating oxidative stress [16-23]. Furthermore, METH induces excitotoxicity by increasing glutamate release, leading to excessive stimulation of postsynaptic receptors and subsequent neuronal cell death [24]. METH exposure has also been strongly associated with neuroinflammation, which further contributes to oxidative stress, promotes excitotoxicity, and disrupts neuroplasticity [25-27] (Fig. 1).
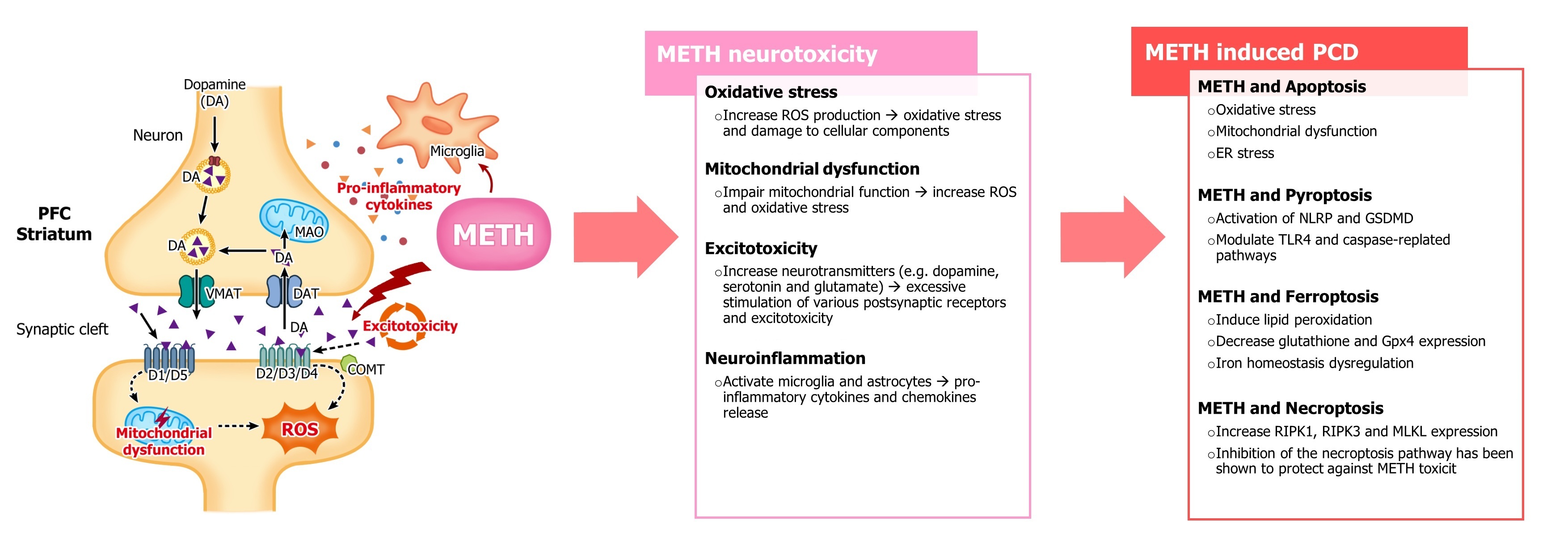
Fig. 1: Molecular mechanisms methamphetamine (METH)-induced neurotoxicity. METH triggers multiple interconnected pathways leading to neuronal programmed cell death (PCD). Oxidative stress, mitochondrial dysfunction, excitotoxicity, and neuroinflammation converge to activate apoptosis, pyroptosis, necroptosis, and ferroptosis. These processes ultimately result in neuronal loss and cognitive impairment. Abbreviations: COMT, Catechol-O-Methyltransferase; DAT, dopamine transporter; MOA, monoamine oxidase; PFC, prefrontal cortex; ROS, reactive oxygen species; VMAT, vesicular monoamine transporter.
Melatonin Against Meth-Induced PCD
Melatonin has been widely recognized for its protective effects against METH-induced neurotoxicity. Its neuroprotective actions are mediated through multiple mechanisms, including direct scavenging of ROS, enhancement of endogenous antioxidant defenses, suppression of inflammatory responses, and modulation of PCD pathways. In addition, melatonin preserves mitochondrial integrity, thereby mitigating mitochondrial dysfunction associated with METH exposure [28]. Among these mechanisms, the antioxidant activity of melatonin represents a key protective property against METH neurotoxicity. METH exposure leads to excessive production of ROS and reactive nitrogen species. Pretreatment with melatonin has been shown to attenuate METH-induced ROS and reactive nitrogen species generation and modulate the TLR4/MyD88/NF-κB signaling pathway [26]. Interestingly, recent evidence further suggests that melatonin may exert neuroprotective effects by modulating ferroptosis, a form of iron-dependent lipid peroxidation-driven PCD that has been increasingly implicated in METH-induced neurotoxicity [29].Molecular Crosstalk Between METH-Induced PCD Pahways and Melatonin-Mediated Neuroprotection
Apoptosis
METH has been shown to induce apoptosis in various cell types [30-33]. The mechanisms of METH-induced apoptosis involve multiple interconnected processes including increased oxidative stress, mitochondrial dysfunction, and ER stress. In particular, METH-induced oxidative stress plays a critical role in promoting apoptotic cell death. The excessive accumulation of ROS subsequently activates pro-apoptotic signaling pathways, disrupts mitochondrial integrity and function, and promotes the release of cytochrome c from mitochondria into the cytosol. This process ultimately triggers the activation of caspase-3 and other downstream apoptotic mediators, leading to PCD [16, 17, 34] (Fig. 2). Mitochondrial dysfunction is a key contributor to apoptosis [21, 35]. METH disrupts mitochondrial integrity by altering mitochondrial structure [22, 36] and inhibiting mitochondrial fusion [37]. In addition, METH induces mitochondrial stress by increasing monoamine oxidase and L-type calcium channel activity [5, 38], while simultaneously reducing the activity of electron transport chain complexes [39]. These damaging factors influences to a decrease in ATP production, thereby releasing pro-apoptotic factors. Collectively, these alterations impair mitochondrial function, resulting in reduced ATP production and the release of pro-apoptotic factors that promote apoptosis (Fig. 2). Moreover, ER stress also contributes to METH-induced apoptosis [40-42]. METH disrupts ER homeostasis by promoting oxidative stress and calcium dysregulation, leading to the accumulation of misfolded proteins and activation of the unfolded protein response (UPR) [41, 43, 44]. These disturbances activate ER stress signaling pathways, including increased expression of PERK and caspase-12 [43]. In addition, C/EBP homologous protein (CHOP), a key regulator of ER stress responses, has been identified as a critical mediator of METH-induced apoptotic signaling [31, 40, 45] (Fig. 2). Regarding interplay between apoptosis and autophagy, METH can either activate or inhibit autophagy, depending on the experimental conditions [10, 21, 46]. Excessive or prolonged autophagy induced by METH may lead to the degradation of mitochondria [18, 21, 22, 47] and lysosomes [48-51]. Furthermore, METH has been reported to decrease Beclin-1 expression while increasing LC3 levels in the brain [31-33, 52-54], suggesting dysregulation of the autophagy pathway. Melatonin has been shown to protect neuronal cells from METH-induced apoptosis by modulating both ER stress and mitochondrial apoptotic signaling pathways. Pretreatment with melatonin attenuates the overexpression of ER stress–related genes and reduces the cleavage of caspase-12 and caspase-3 [55]. Moreover, prior administration of melatonin can reverse the effect of METH-induced apoptosis by increasing the Bcl-XL/Bax ratio, reducing cytochrome c release, and decreasing the cleavage of caspase-9 and caspase-3 [56]. Melatonin can reduce METH-induced neurotoxicity by regulating autophagy [57]. Specifically, melatonin enhances mTOR signaling and promotes the phosphorylation of the eukaryotic initiation factor 4E-binding protein 1 (4EBP1), thereby suppressing LC3-II formation [58]. Furthermore, melatonin inhibits autophagy-related cell death through modulation of the Bcl-2/Beclin-1 pathway, partly by suppressing JNK1 activation [59]. Altogether, these findings indicate that melatonin exerts neuroprotective effects by regulating both apoptotic and autophagic pathways involved in METH-induced neuronal injury (Fig. 2).
Fig. 2: PCD signaling pathways in METH-induced neurotoxicity and the multifaceted neuroprotective mechanisms of melatonin. Clockophagy refers to a specific type of selective autophagy that targets and degrades core proteins of the circadian clock.
Pyroptosis
Pyroptosis is an inflammatory form of PCD characterized by the formation of membrane pores, release of pro-inflammatory cytokines, and activation of the inflammasome. Substantial evidence indicates that METH induces pyroptosis primarily through activation of the NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome, which mediates the cleavage and release of pro-inflammatory cytokines such as interleukin-1β (IL-1β) and interleukin-18 (IL-18) [60, 61]. METH has also been reported to induce Gasdermin E (GSDME) expression in the mouse hippocampus, a process associated with ER stress signaling [43, 62]. The toxicity METH may involve the NLRP1–caspase-1–GSDMD signaling pathway, and inhibition of NLRP1 has been shown to attenuate METH-induced dysfunction [63]. Other molecular pathways, including Toll-like receptor 4 (TLR4) signaling [54, 64-68] and the caspase-11 pathway [63] have also been implicated. These pathways ultimately converge on the activation of gasdermin proteins, particularly GSDMD, leading to pyroptotic cell death (Fig. 2). Accumulating evidence indicates that melatonin can counteract pyroptosis primarily by inhibiting activation of the NLRP3 inflammasome [69, 70]. For example, melatonin attenuates lipopolysaccharide (LPS)-induced depressive-like behavior in mice by suppressing ATP-mediated NLRP3 activation in microglia. This protective effect is associated with activation of nuclear factor erythroid 2–related factor 2 (Nrf2) and silent information regulator 2 homolog 1 (SIRT1) signaling pathways [71]. Furthermore, melatonin significantly diminished neuronal injury in streptozotocin-induced diabetic mice and in neuronal tissue exposed to high glucose levels by inhibiting pyroptosis and excessive autophagy activation [72]. Through suppression of the NF-κB/NLRP3 signaling axis, melatonin also alleviates inflammatory responses in multiple tissues, including the aortic endothelium [73], retinal neurons [74, 75], liver [76, 77], cardiomyocyte [78-80], spinal nerve [81], and kidney [82]. These protective effects are further supported by several regulatory pathways involving SIRT1, microRNAs, long non-coding RNAs, and Wnt/β-catenin signaling [69, 71, 83] (Fig. 2).
Necroptosis
Necroptosis is a lytic form of PCD characterized by loss of plasma membrane integrity, organelle swelling, and release of intracellular contents. This process is mediated by activation of receptor-interacting protein kinase 1 (RIPK1) and RIPK3, which form a necrosome complex. The necrosome subsequently activates mixed lineage kinase domain-like (MLKL), leading to membrane permeabilization and necroptotic cell death [84]. Necroptosis and its molecular mediators have been implicated in several central nervous system disorders, including amyloid-β–associated pathology in Alzheimer’s disease [85, 86], Parkinson's disease [87], viral infections of the CNS [88, 89], cerebral ischemia [90, 91], and glutamate-induced retinal ganglion cell loss in glaucoma models [92] (Fig. 2). Evidence also suggests that METH exposure can trigger necroptosis. METH has been reported to increase the expression of RIPK1 [93] and RIPK3 [94, 95]. Inhibition of RIPK1 with necrostatin-1 has been shown to protect against METH-induced neurotoxicity, as demonstrated by improved behavioral performance and reduced neuronal damage in the striatum of mice [93]. Additionally, METH exposure increases MLKL expression and induces necroptosis in microglial cells [94, 96]. These findings indicate that necroptosis contribute to METH-induced neurotoxicity. Therefore, targeting the necroptosis signaling pathway may represent a potential therapeutic strategy for mitigating METH-induced neuronal damage, although further studies are required to clarify the underlying mechanisms. Melatonin has been reported to suppress necroptotic signaling in various experimental models. For instance, melatonin alleviates chronic lung inflammation while reducing the expression of RIPK1, RIPK3, and MLKL [97, 98]. It also inhibits RIPK3-mediated necroptosis through regulation of the deubiquitinating enzyme A20, also known as tumor necrosis factor alpha–induced protein 3, TNFAIP3 [98, 99]. Furthermore, melatonin attenuates oxidative stress and inflammation, thereby preventing formation of the RIPK1–RIPK3 complex in liver fibrosis models [98, 100]. In concert, the anti-necroptotic effects of melatonin involve suppression of the RIPK1/RIPK3/MLKL signaling axis, modulation of A20, and inhibition of inflammatory DAMP–TLR4–HMGB1 pathways (Fig. 2).
Meth, Melatonin, And Ferroptosis: A Circadian Perspective on Neurotoxicity and Neuroprotection
Crosstalk Among Apoptosis, Pyroptosis, And Necroptosis in Panoptosis PANoptosis is an inflammatory PCD pathway that integrates the features of apoptosis, pyroptosis, and necroptosis into a coordinated process. It is uniquely characterized by the assembly of a multi-protein complex known as the PANoptosome, which acts as a molecular scaffold to initiate these three pathways [101]. Unlike traditional cell death models that view these processes as isolated events, PANoptosis highlights their functional redundancy and crosstalk, allowing cell death to proceed even if one pathway is inhibited (e.g., by viral inhibitors). The mechanism is primarily driven by specific sensors like ZBP1 or AIM2, which recruit adaptors like ASC and FADD to activate catalytic effectors including caspases-1, -8, and RIPK1/3 [102, 103]. When PANoptosis is overactivated, it triggers a massive release of pro-inflammatory cytokines leading to extensive tissue damage and cytokine storms observed in conditions like COVID-19 [104-106], sepsis [107], and neurodegenerative disorders [101, 108-111]. Melatonin and METH exert opposed effects on PANoptosis. METH acts as a potent inducer of PANoptotic signaling, as evidenced by its ability to trigger apoptosis, activate pyroptosis, and promote necroptosis. In contrast, melatonin functions as a cytoprotective agent, attenuating apoptosis and necroptosis while counteracting PANoptosome activation through suppression of the melatonin receptor 2 (MT2)/sirtuin 1 (SIRT1)/E26 oncogene homolog 1 (ETS1)/NLRP3 signaling axis [112, 113].
Ferroptosis
Ferroptosis is a form of regulated cell death driven by iron-dependent lipid peroxidation, resulting in lethal accumulation of lipid peroxides and subsequent cellular damage. This process is typically associated with depletion of intracellular glutathione (GSH), inhibition of cystine transport through the system Xc⁻ transporter, and inactivation of glutathione peroxidase 4 (GPX4), leading to excessive lipid peroxidation (Fig. 2). Recent studies have suggested a potential link between METH exposure and ferroptosis. Although the precise mechanisms remain unclear, previous evidence indicates that METH can induce ferroptotic processes in the brain. In vitro studies demonstrate that METH promotes lipid peroxidation, a hallmark of ferroptosis [3, 34, 114-116]. Additionally, METH has been reported to reduce the expression of GPX4, a key antioxidant enzyme that protects cells from lipid peroxidation and ferroptotic cell death [3, 117, 118]. Taken together, these findings suggest that ferroptosis may contribute to METH-induced neurotoxicity, although further studies are needed to clarify the underlying molecular mechanisms. Several studies suggest that melatonin may serve as a promising anti-ferroptotic agent. Melatonin can inhibit ferroptosis by enhancing antioxidant defense systems, particularly through activation of the Nrf2 and GPX4 pathways. In addition, melatonin regulates iron homeostasis, thereby limiting the accumulation of excess intracellular iron that promotes lipid peroxidation. Experimental evidence supports the protective role of melatonin against ferroptosis in the nervous system. For instance, melatonin suppresses ferroptosis in a mouse model of acute sleep deprivation [119] and prevents hypoxic–ischemic hippocampal injury in rats [120]. These studies suggest that melatonin exerts its anti-ferroptotic effects largely through activation of the Akt/Nrf2/GPX4 signaling pathway. Beyond the nervous system, melatonin has also been shown to inhibit ferroptosis in several pathological conditions by regulating Nrf2-related pathways. These include models of osteoporosis [121-123], PM2.5-induced lung injury [124], COVID-19–associated disruption of iron–redox homeostasis [125], and acute kidney injury [122, 126]. In sum, these findings highlight the potential of melatonin as a broad-spectrum inhibitor of ferroptosis (Fig. 2). In addition to its direct free radical scavenging activity, melatonin also acts as an iron chelator and can inhibit lipid peroxidation induced by iron overload [127]. Melatonin has been reported to suppress the expression of transferrin receptor 1 (TfR1), a key protein involved in iron uptake, while promoting the expression of ferritin, which is responsible for iron storage. These actions reduce intracellular iron levels, thereby limiting lipid peroxidation and ferroptosis [119]. In a murine model of age-related cataract, melatonin suppressed ferroptosis by modulating the SIRT6/phosphorylated-Nrf2/GPX4 and SIRT6/NCOA4/ferritin heavy chain 1 pathways [128]. Moreover, melatonin exerts neuroprotective effects in traumatic brain injury by inhibiting neuronal ferritin heavy chain–mediated ferroptosis. Similarly, N-acetylserotonin, the immediate precursor of melatonin, has been reported to alleviate traumatic brain injury through activation of the Nrf2/ferritin heavy chain 1 pathway [129]. Although these findings highlight the anti-ferroptotic potential of melatonin, its role in METH-induced ferroptosis remains largely unexplored and warrants further investigation.
Meth, Melatonin, And Ferroptosis: A Circadian Perspective on Neurotoxicity and Neuroprotection
Circadian rhythms are endogenous biological oscillations that regulate numerous physiological processes, including metabolism, redox balance, and neuronal function. These rhythms are controlled by molecular clock genes such as CLOCK, PER, CRY, and ARNTL (BMAL1), which coordinate cellular homeostasis across the 24-hour cycle. Disruption of circadian regulation has been increasingly recognized as an important factor contributing to oxidative stress, neuroinflammation, and neuronal vulnerability in various neurological disorders. Both METH and melatonin are capable of entraining circadian rhythms in the nervous system [130]. The circadian clock may serve as an important mediator through which METH or melatonin regulates cell death pathways. Molecular interactions between circadian clock components and ferroptosis are increasingly recognized as bidirectional and closely intertwined. A specialized form of selective autophagy, termed clockophagy, promotes ferroptosis by targeting the core circadian regulator BMAL1 for degradation via the cargo receptor SQSTM1/p62, thereby enhancing lipid peroxidation and ferroptotic cell death, particularly in cancer cells [131, 132] [133]. In addition, BMAL1 directly regulates the expression of GPX4, a key suppressor of ferroptosis. Disruption of BMAL1 in renal cells exposed to toxic stimuli abolishes the normal rhythmic expression of GPX4, resulting in increased lipid peroxidation and heightened susceptibility to ferroptotic cell death [134]. Furthermore, in models of kidney ischemia–reperfusion injury, BMAL1 regulates ferroptosis via the NRF2 antioxidant pathway, and loss of BMAL1 reduces NRF2 activity, thereby exacerbating oxidative stress and promoting ferroptotic tissue damage [135]. Similarly, in sepsis-induced myocardial injury, decreased BMAL1 expression alters AKT/p53 signaling, shifting the balance toward cell death and promoting ferroptosis [136]. These findings highlight the critical role of circadian clock components in regulating redox homeostasis and temporally controlling ferroptosis. In human hepatocellular carcinoma cells (HepG2) and mouse primary hepatocytes, melatonin has been shown to protect against ethanol-induced ferroptosis by promoting nuclear translocation of NRF2 and activating downstream anti-ferroptotic targets, including ferritin heavy chain (FTH), ferroportin (FPN), heme oxygenase-1 (HO-1), and SLC7A1. However, knockdown of BMAL1, which disrupts the NRF2–ARE signaling pathway, abolishes the anti-ferroptotic effects of melatonin in the liver. These findings indicate that the integrity of the circadian clock is essential for melatonin-mediated resistance to ferroptosis [137]. Other circadian clock genes also participate in the regulation of ferroptosis. The nuclear receptor subfamily 1 group D member 1 (REV-ERB) is another clock component implicated in ferroptosis control. Loss of REV-ERBα or REV-ERBβ has been shown to reduce susceptibility to folic acid–induced acute kidney injury in mice. Mechanistically, these transcription factors promote ferroptosis by repressing the transcription of anti-ferroptotic genes, including SLC7A11 and HO-1, through direct binding to the Rev-ERB response element (RORE) in their promoter regions [138]. Conversely, pharmacological activation of REV-ERB with the agonist SR9009 has been reported to alleviate ischemia–reperfusion injury by suppressing ferroptosis in cardiomyocytes of diabetic rats [139]. In addition to REV-ERB, Period (PER) proteins, which are core components of the circadian clock, has been associated with the regulation of ferroptosis as well as the progression of several malignancies. For instance, Yang et al. (2022) reported that PER1 expression was negatively correlated with GPX4 and hypoxia-inducible factor-1α (HIF-1α) levels in oral squamous cell carcinoma tissues [140]. Furthermore, in a model of nonalcoholic steatohepatitis, PER2 knockout suppressed ferroptosis by reducing lipid peroxidation, downregulating ferroptosis-related genes, and improving mitochondrial morphology in hepatocytes. These findings suggest that PER2 contributes to the regulation and promotion of ferroptotic processes [141]. METH exposure is known to disrupt circadian rhythms and alter the expression of core clock genes in the brain. Such circadian disturbances can exacerbate oxidative stress, mitochondrial dysfunction, and iron dysregulation, which is key processes involved in ferroptotic cell death. Experimental evidence indicates that reduced BMAL1 expression increases neuronal susceptibility to ferroptotic injury, whereas restoration of circadian signaling can confer neuroprotection [142]. Conversely, melatonin has emerged as a potent inhibitor of ferroptosis with potential therapeutic implications for neurodegenerative conditions, including post-COVID-19 neurological complications [143]. Melatonin exerts neuroprotective effects through multiple mechanisms, including direct scavenging of reactive oxygen species, regulation of iron metabolism, and activation of antioxidant pathways such as Nrf2 and GPX4, which are critical regulators of ferroptosis (Fig. 3). Melatonin can also suppress inflammatory signaling and improve mitochondrial function. In sum, these pleiotropic actions suggest that melatonin may counteract ferroptosis-associated neuronal injury and neurodegeneration, highlighting its therapeutic potential in mitigating long-term neurological damage. Emerging evidence indicates that the circadian clock–melatonin–ferroptosis axis represents an important regulatory network in neurodegeneration. In the context of METH exposure, circadian disruption may exacerbate ferroptotic processes, whereas melatonin may attenuate these effects by restoring circadian homeostasis and enhancing antioxidant defenses. Elucidating this circadian–ferroptosis interplay may provide new insights into the mechanisms of METH-induced neurotoxicity and reveal novel targets for neuroprotective interventions (Fig. 3).
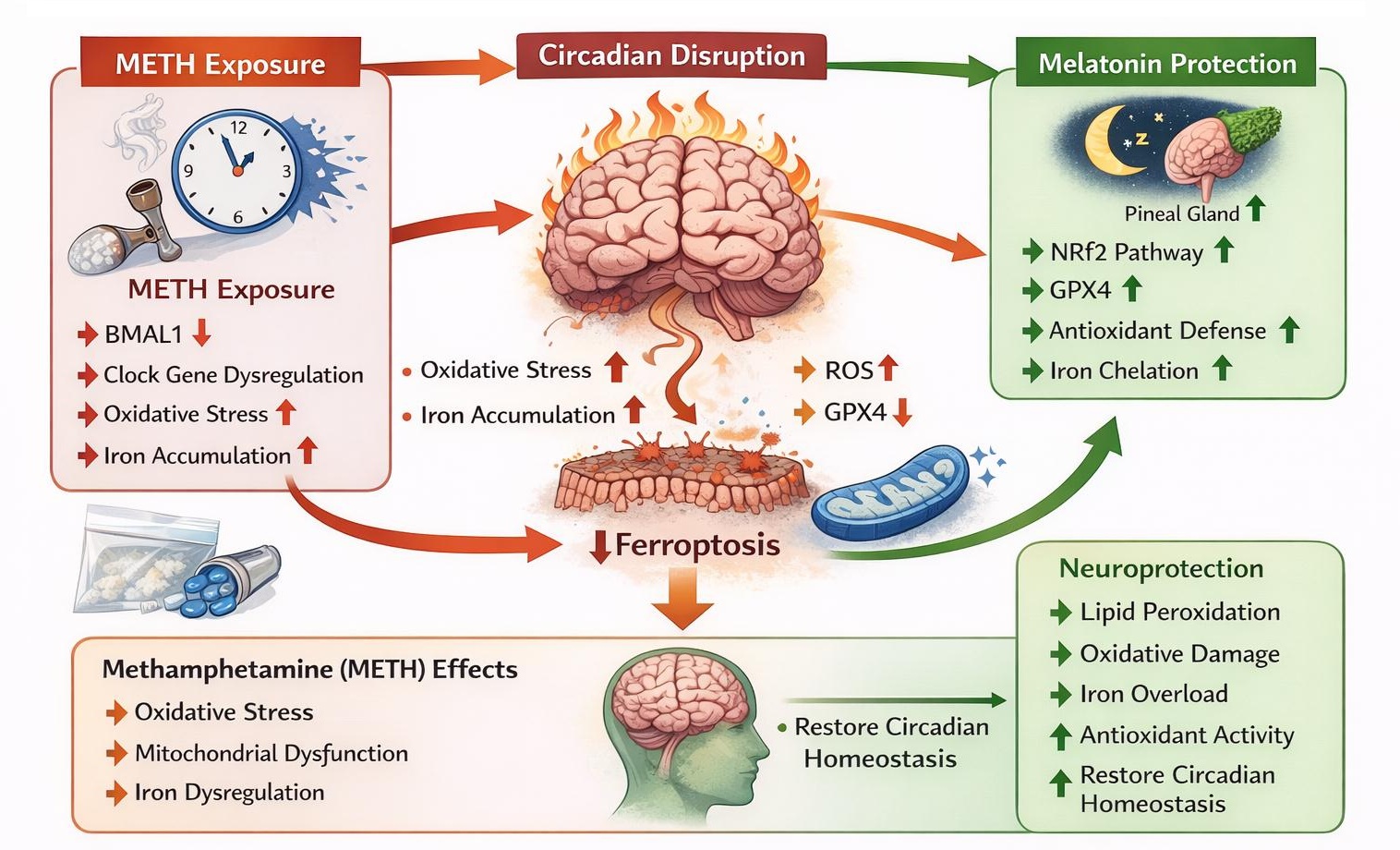
Fig. 3: Circadian clock-ferroptosis-melatonin-METH axis in neuroprotection. METH exposure disrupts circadian rhythm regulation in the brain by altering the expression of core clock genes, particularly BMAL1, leading to circadian misalignment. Circadian disruption enhances oxidative stress, mitochondrial dysfunction, and iron accumulation, which together promote lipid peroxidation and trigger ferroptotic neuronal cell death. Impairment of antioxidant systems, including GPX4, further increases neuronal susceptibility to ferroptosis. Melatonin, the primary circadian hormone secreted by the pineal gland, counteracts these pathological processes through multiple protective mechanisms. Melatonin restores circadian homeostasis by regulating BMAL1 signaling, activates the Nrf2 antioxidant pathway, enhances GPX4 activity, reduces ROS generation, and limits iron-dependent lipid peroxidation. Through these coordinated actions, melatonin suppresses ferroptotic signaling and protects neurons from METH-induced neurotoxicity. This circadian clock–melatonin–ferroptosis regulatory axis highlights a potential therapeutic target for mitigating METH-related neuronal injury and neurodegeneration.
Limitations And Future Perspective
Despite compelling evidence supporting the neuroprotective effects of melatonin against METH-induced neurotoxicity, several important limitations remain. First, the majority of available data are derived from preclinical models, predominantly rodents and in vitro systems, which may not fully recapitulate the complexity of human METH abuse, including variability in dosing patterns, duration of exposure, and comorbid conditions. Second, while melatonin’s ability to modulate multiple PCD pathways is well documented, the precise temporal hierarchy and causal relationships among apoptosis, pyroptosis, necroptosis, and ferroptosis during METH-induced neurodegeneration remain insufficiently defined. In particular, the emerging circadian–ferroptosis axis, though conceptually compelling, is supported by limited mechanistic evidence in the context of METH neurotoxicity, and direct causal links between circadian disruption, ferroptosis activation, and melatonin-mediated protection require further validation. Additionally, optimal dosing regimens, treatment duration, and timing of melatonin administration relative to METH exposure are poorly characterized, and long-term efficacy and safety in the context of chronic METH use remains largely unexplored. Future research should focus on translating these preclinical findings into clinically relevant frameworks. Controlled human clinical trials are urgently needed to evaluate melatonin supplementation during METH withdrawal and recovery, with attention to cognitive, behavioral, and neurobiological outcomes. Time-resolved studies integrating circadian biology with PCD pathway activation will be essential to clarify whether circadian misalignment serves as a driver or amplifier of ferroptosis and other death mechanisms in METH-induced neurotoxicity. The development of reliable biomarkers reflecting oxidative stress, ferroptosis activity, and circadian disruption could facilitate treatment stratification and therapeutic monitoring. Furthermore, investigation of melatonin analogs or formulations with improved blood–brain barrier penetration may enhance therapeutic efficacy. Finally, aligning melatonin administration with endogenous circadian rhythms may maximize neuroprotection and improve clinical outcomes. A physiological-dose formulation (0.3–1.0 mg), designed to mimic the natural dim-light melatonin profile together with appropriately timed and sustained administration may restore circadian homeostasis [144-146], thereby enhancing antioxidant capacity and reducing susceptibility to ferroptosis, supporting its potential as a chronotherapeutic intervention. Addressing current limitations will be essential to advance melatonin from a promising neuroprotective agent to an evidence-based therapy for mitigating the neurological consequences of METH abuse.
Conclusion
This comprehensive review highlights the robust neuroprotective potential of melatonin against METH-induced neurotoxicity through the modulation of multiple PCD pathways. Evidence indicates that melatonin attenuates apoptosis, pyroptosis, necroptosis, and ferroptosis through interconnected mechanisms, mainly by suppressing oxidative stress, mitochondrial dysfunction, and neuroinflammation, which are key upstream drivers of neuronal injury. Emerging findings also suggest a concept of circadian–ferroptosis axis, in which circadian clock regulation may link the chronobiotic and neuroprotective actions of melatonin. Given its well-established safety profile and multi-target protective properties, melatonin represents a promising candidate for therapeutic intervention in METH-related neurological damage. These findings underscore the imperative for rigorously designed clinical studies to translate preclinical evidence into effective therapeutic strategies aimed at attenuating the neurobiological sequelae of METH exposure.
Acknowledgements
The authors used AI assistance (ChatGPT) solely to improve the language and grammar of the manuscript. Fig. 3 was generated using AI. All content was subsequently reviewed and verified by the authors, who take full responsibility for the accuracy and integrity of the work.
Author Contributions N.P. and A.J. conceived the idea. N.P., S.L., P.T., and P.N. drafted the abstract and the review manuscript. P.T. and A.J. prepared the original figures and diagrams. N.P., P.T., and O.T. edited and validated the manuscript. All authors have read and approved the final version of the manuscript.
Funding Sources This research project is supported by Chulabhorn Royal Academy (Fundamental Fund by National Science and Research Innovation Fund (NSRF): fiscal year 2024) (FRB670044/0240 Project code198491).
Disclosure Statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
- Moujalled D, Strasser A, Liddell JR: Molecular mechanisms of cell death in neurological diseases. Cell Death Differ 2021;28:2029-2044.https://doi.org/xxxx10.1038/s41418-021-00814-y
- Guo D, Liu Z, Zhou J, Ke C, Li D: Significance of Programmed Cell Death Pathways in Neurodegenerative Diseases. Int J Mol Sci 2024;25https://doi.org/xxxx10.3390/ijms25189947
- Guo D, Huang X, Xiong T, Wang X, Zhang J, Wang Y, Liang J: Molecular mechanisms of programmed cell death in methamphetamine-induced neuronal damage. Front Pharmacol 2022;13:980340.https://doi.org/xxxx10.3389/fphar.2022.980340
- Berro LF, Overton JS, Rowlett JK: Methamphetamine-Induced Sleep Impairments and Subsequent Slow-Wave and Rapid Eye Movement Sleep Rebound in Male Rhesus Monkeys. Front Neurosci 2022;16:866971.https://doi.org/xxxx10.3389/fnins.2022.866971
- Du Y, Lee YB, Graves SM: Chronic methamphetamine-induced neurodegeneration: Differential vulnerability of ventral tegmental area and substantia nigra pars compacta dopamine neurons. Neuropharmacology 2021;200:108817.https://doi.org/xxxx10.1016/j.neuropharm.2021.108817
- Jayanthi S, Daiwile AP, Cadet JL: Neurotoxicity of methamphetamine: Main effects and mechanisms. Exp Neurol 2021;344:113795.https://doi.org/xxxx10.1016/j.expneurol.2021.113795
- Omidvari S, Azimzadeh Z, Rashnoo F, Tahmasebinia F, Keramatinia A, Roozbahany NA, Abbaszadeh HA, Darabi S: Molecular mechanisms and treatment strategies for methamphetamine‑induced neurodegeneration, inflammation and neurotoxicity. Acta Neurobiol Exp (Wars) 2023;83:414-431.https://doi.org/xxxx10.55782/ane-2023-2488
- Cheng YJ, Deng YZ, Deng D, Wu MQ, Chai JR, Wang YJ, Liu JG, Zhao M: Prelimbic cortex dynorphin/κ opioid receptor system modulates methamphetamine-induced cognitive impairment. Addict Biol 2023;28:e13323.https://doi.org/xxxx10.1111/adb.13323
- Wang J, Hao Y, Ma D, Feng L, Yang F, An P, Su X, Feng J: Neurotoxicity mechanisms and clinical implications of six common recreational drugs. Front Pharmacol 2025;16:1526270.https://doi.org/xxxx10.3389/fphar.2025.1526270
- Shrestha P, Katila N, Lee S, Seo JH, Jeong JH, Yook S: Methamphetamine induced neurotoxic diseases, molecular mechanism, and current treatment strategies. Biomed Pharmacother 2022;154:113591.https://doi.org/xxxx10.1016/j.biopha.2022.113591
- Gibson AS, West PJ, Keefe KA: Effects of methamphetamine-induced neurotoxicity on striatal long-term potentiation. Psychopharmacology (Berl) 2022;239:93-104.https://doi.org/xxxx10.1007/s00213-021-06055-8
- Hosseini A, Shetab-Boushehri SM, Shetab-Boushehri SV: Evaluation of Cytotoxic, Necrotic, Apoptotic, and Autophagic Effects of Methamphetamine and 3, 4-Methylenedioxymethamphetamine on U-87 MG (Glial) and B104-1-1 (Neuronal) Cell Lines. Neurotox Res 2022;40:1499-1515.https://doi.org/xxxx10.1007/s12640-022-00543-1
- Karimi-Haghighi S, Chavoshinezhad S, Mozafari R, Noorbakhsh F, Borhani-Haghighi A, Haghparast A: Neuroinflammatory Response in Reward-Associated Psychostimulants and Opioids: A Review. Cell Mol Neurobiol 2023;43:649-682.https://doi.org/xxxx10.1007/s10571-022-01223-6
- Miller AE, Daiwile AP, Cadet JL: Sex-Dependent Alterations in the mRNA Expression of Enzymes Involved in Dopamine Synthesis and Breakdown After Methamphetamine Self-Administration. Neurotox Res 2022;40:1464-1478.https://doi.org/xxxx10.1007/s12640-022-00545-z
- Persons JE, Conway KS: Neuropathologic Features in Chronic Methamphetamine Use. Am J Forensic Med Pathol 2023https://doi.org/xxxx10.1097/PAF.0000000000000817
- Peirouvi T, Razi M: Molecular mechanism behind methamphetamine-induced damages in testicular tissue: Evidences for oxidative stress, autophagy, and apoptosis. Andrologia 2022;54:e14534.https://doi.org/xxxx10.1111/and.14534
- Xu H, Jiang C, Zhao H, Liu L: 6-Formyl-5-isopropyl-3-hydroxymethyl- 7-methyl-1H-indene mitigates methamphetamine-induced photoreceptor cell toxicity through inhibiting oxidative stress. Hum Exp Toxicol 2020;39:712-720.https://doi.org/xxxx10.1177/0960327119896617
- Abdullah CS, Remex NS, Aishwarya R, Nitu S, Kolluru GK, Traylor J, Hartman B, King J, Bhuiyan MAN, Hall N, Murnane KS, Goeders NE, Kevil CG, Orr AW, Bhuiyan MS: Mitochondrial dysfunction and autophagy activation are associated with cardiomyopathy developed by extended methamphetamine self-administration in rats. Redox Biol 2022;58:102523.https://doi.org/xxxx10.1016/j.redox.2022.102523
- Zeng J, Tao J, Xia L, Zeng Z, Chen J, Wang Z, Meng J, Liu L: Melatonin inhibits vascular endothelial cell pyroptosis by improving mitochondrial function via up-regulation and demethylation of UQCRCBiochem Cell Biol 2021;99:339-347.https://doi.org/xxxx10.1139/bcb-2020-0279
- Sun X, Sun P, Zhen D, Xu X, Yang L, Fu D, Wei C, Niu X, Tian J, Li H: Melatonin alleviates doxorubicin-induced mitochondrial oxidative damage and ferroptosis in cardiomyocytes by regulating YAP expression. Toxicol Appl Pharmacol 2022;437:115902.https://doi.org/xxxx10.1016/j.taap.2022.115902
- Ryskalin L, Biagioni F, Busceti CL, Polzella M, Lenzi P, Frati A, Ferrucci M, Fornai F: Lactoferrin Protects against Methamphetamine Toxicity by Modulating Autophagy and Mitochondrial Status. Nutrients 2021;13https://doi.org/xxxx10.3390/nu13103356
- Lenzi P, Biagioni F, Busceti CL, Lazzeri G, Polzella M, Frati A, Ferrucci M, Fornai F: Alterations of Mitochondrial Structure in Methamphetamine Toxicity. Int J Mol Sci 2022;23https://doi.org/xxxx10.3390/ijms23168926
- Turan Ç, Şenormancı G, Neşelioğlu S, Budak Y, Erel Ö, Şenormancı Ö: Oxidative Stress and Inflammatory Biomarkers in People with Methamphetamine Use Disorder. Clin Psychopharmacol Neurosci 2023;21:572-582.https://doi.org/xxxx10.9758/cpn.22.1047
- Nicosia N, Giovenzana M, Misztak P, Mingardi J, Musazzi L: Glutamate-Mediated Excitotoxicity in the Pathogenesis and Treatment of Neurodevelopmental and Adult Mental Disorders. Int J Mol Sci 2024;25https://doi.org/xxxx10.3390/ijms25126521
- Hein ZM, Kraiwattanapirom N, Mukda S, Chetsawang B: The induction of Neuron-Glial2 (NG2) expressing cells in methamphetamine toxicity-induced neuroinflammation in rat brain are averted by melatonin. J Neuroimmunol 2020;344:577232.https://doi.org/xxxx10.1016/j.jneuroim.2020.577232
- Lwin T, Yang JL, Ngampramuan S, Viwatpinyo K, Chancharoen P, Veschsanit N, Pinyomahakul J, Govitrapong P, Mukda S: Melatonin ameliorates methamphetamine-induced cognitive impairments by inhibiting neuroinflammation via suppression of the TLR4/MyD88/NFκB signaling pathway in the mouse hippocampus. Prog Neuropsychopharmacol Biol Psychiatry 2021;111:110109.https://doi.org/xxxx10.1016/j.pnpbp.2020.110109
- Chen L, Ru Q, Xiong Q, Yang J, Xu G, Wu Y: Potential Effects of Nrf2 in Exercise Intervention of Neurotoxicity Caused by Methamphetamine Oxidative Stress. Oxid Med Cell Longev 2022;2022:4445734.https://doi.org/xxxx10.1155/2022/4445734
- Polvat T, Prasertporn T, Na Nakorn P, Pannengpetch S, Suwanjang W, Panmanee J, Ngampramuan S, Cornish JL, Chetsawang B: Proteomic Analysis Reveals the Neurotoxic Effects of Chronic Methamphetamine Self-Administration-Induced Cognitive Impairments and the Role of Melatonin-Enhanced Restorative Process during Methamphetamine Withdrawal. J Proteome Res 2023;22:3348-3359.https://doi.org/xxxx10.1021/acs.jproteome.3c00502
- Rui T, Wang H, Li Q, Cheng Y, Gao Y, Fang X, Ma X, Chen G, Gao C, Gu Z, Song S, Zhang J, Wang C, Wang Z, Wang T, Zhang M, Min J, Chen X, Tao L, Wang F, et al.: Deletion of ferritin H in neurons counteracts the protective effect of melatonin against traumatic brain injury-induced ferroptosis. J Pineal Res 2021;70:e12704.https://doi.org/xxxx10.1111/jpi.12704
- Foroughi K, Jahanbani S, Khaksari M, Shayannia A: Obestatin attenuated methamphetamine-induced PC12 cells neurotoxicity via inhibiting autophagy and apoptosis. Hum Exp Toxicol 2020;39:301-310.https://doi.org/xxxx10.1177/0960327119886036
- Tabatabaei Mirakabad FS, Khoramgah MS, Abdollahifar MA, Tehrani AS, Rezaei-Tavirani M, Niknazar S, Tahmasebinia F, Mahmoudiasl GR, Khoshsirat S, Abbaszadeh HA: NUPR1- CHOP experssion, autophagosome formation and apoptosis in the postmortem striatum of chronic methamphetamine user. J Chem Neuroanat 2021;114:101942.https://doi.org/xxxx10.1016/j.jchemneu.2021.101942
- Tan XH, Zhang KK, Xu JT, Qu D, Chen LJ, Li JH, Wang Q, Wang HJ, Xie XL: Luteolin alleviates methamphetamine-induced neurotoxicity by suppressing PI3K/Akt pathway-modulated apoptosis and autophagy in rats. Food Chem Toxicol 2020;137:111179.https://doi.org/xxxx10.1016/j.fct.2020.111179
- Zhang KK, Wang H, Qu D, Chen LJ, Wang LB, Li JH, Liu JL, Xu LL, Yoshida JS, Xu JT, Xie XL, Li DR: Luteolin Alleviates Methamphetamine-Induced Hepatotoxicity by Suppressing the p53 Pathway-Mediated Apoptosis, Autophagy, and Inflammation in Rats. Front Pharmacol 2021;12:641917.https://doi.org/xxxx10.3389/fphar.2021.641917
- Lee C, Jang JH, Park GH: α-Pinene Attenuates Methamphetamine-Induced Conditioned Place Preference in C57BL/6 Mice. Biomol Ther (Seoul) 2023https://doi.org/xxxx10.4062/biomolther.2022.132
- Chen X, Qiu F, Zhao X, Lu J, Tan X, Xu J, Chen C, Zhang F, Liu C, Qiao D, Wang H: Astrocyte-Derived Lipocalin-2 Is Involved in Mitochondrion-Related Neuronal Apoptosis Induced by Methamphetamine. ACS Chem Neurosci 2020;11:1102-1116.https://doi.org/xxxx10.1021/acschemneuro.9b00559
- Ferrucci M, Busceti CL, Lazzeri G, Biagioni F, Puglisi-Allegra S, Frati A, Lenzi P, Fornai F: Bacopa Protects against Neurotoxicity Induced by MPP(+) and Methamphetamine. Molecules 2022;27https://doi.org/xxxx10.3390/molecules27165204
- Wang L, Wei Q, Xu R, Chen Y, Li S, Bu Q, Zhao Y, Li H, Zhao Y, Jiang L, Chen Y, Dai Y, Zhao Y, Cen X: Cardiolipin and OPA1 Team up for Methamphetamine-Induced Locomotor Activity by Promoting Neuronal Mitochondrial Fusion in the Nucleus Accumbens of Mice. ACS Chem Neurosci 2023;14:1585-1601.https://doi.org/xxxx10.1021/acschemneuro.2c00709
- Du Y, Choi S, Pilski A, Graves SM: Differential vulnerability of locus coeruleus and dorsal raphe neurons to chronic methamphetamine-induced degeneration. Front Cell Neurosci 2022;16:949923.https://doi.org/xxxx10.3389/fncel.2022.949923
- Bazylianska V, Sharma A, Chauhan H, Schneider B, Moszczynska A: Dopamine and Methamphetamine Differentially Affect Electron Transport Chain Complexes and Parkin in Rat Striatum: New Insight into Methamphetamine Neurotoxicity. Int J Mol Sci 2021;23https://doi.org/xxxx10.3390/ijms23010363
- Yang L, Guo N, Fan W, Ni C, Huang M, Bai L, Zhang L, Zhang X, Wen Y, Li Y, Zhou X, Bai J: Thioredoxin-1 blocks methamphetamine-induced injury in brain through inhibiting endoplasmic reticulum and mitochondria-mediated apoptosis in mice. Neurotoxicology 2020;78:163-169.https://doi.org/xxxx10.1016/j.neuro.2020.03.006
- Zeng Y, Chen Y, Zhang S, Ren H, Xia J, Liu M, Shan B, Ren Y: Natural Products in Modulating Methamphetamine-Induced Neuronal Apoptosis. Front Pharmacol 2021;12:805991.https://doi.org/xxxx10.3389/fphar.2021.805991
- Chen X, Shi C, He M, Xiong S, Xia X: Endoplasmic reticulum stress: molecular mechanism and therapeutic targets. Signal Transduct Target Ther 2023;8:352.https://doi.org/xxxx10.1038/s41392-023-01570-w
- Liu Y, Wen D, Gao J, Xie B, Yu H, Shen Q, Zhang J, Jing W, Cong B, Ma C: Methamphetamine induces GSDME-dependent cell death in hippocampal neuronal cells through the endoplasmic reticulum stress pathway. Brain Res Bull 2020;162:73-83.https://doi.org/xxxx10.1016/j.brainresbull.2020.06.005
- Chen G, Yu G, Yong Z, Yan H, Su R, Wang H: A large dose of methamphetamine inhibits drug‑evoked synaptic plasticity via ER stress in the hippocampus. Mol Med Rep 2021;23https://doi.org/xxxx10.3892/mmr.2021.11917
- Tan X, Cai D, Chen N, Du S, Qiao D, Yue X, Wang T, Li J, Xie W, Wang H: Methamphetamine mediates apoptosis of vascular smooth muscle cells via the chop-related endoplasmic reticulum stress pathway. Toxicol Lett 2021;350:98-110.https://doi.org/xxxx10.1016/j.toxlet.2021.06.019
- Limanaqi F, Busceti CL, Celli R, Biagioni F, Fornai F: Autophagy as a gateway for the effects of methamphetamine: From neurotransmitter release and synaptic plasticity to psychiatric and neurodegenerative disorders. Prog Neurobiol 2021;204:102112.https://doi.org/xxxx10.1016/j.pneurobio.2021.102112
- Soares J, Costa VM, Gaspar H, Santos S, Bastos ML, Carvalho F, Capela JP: Adverse outcome pathways induced by 3, 4-dimethylmethcathinone and 4-methylmethcathinone in differentiated human SH-SY5Y neuronal cells. Arch Toxicol 2020;94:2481-2503.https://doi.org/xxxx10.1007/s00204-020-02761-y
- Barbaro JM, Sidoli S, Cuervo AM, Berman JW: Methamphetamine Dysregulates Macrophage Functions and Autophagy to Mediate HIV Neuropathogenesis. Biomedicines 2022;10https://doi.org/xxxx10.3390/biomedicines10061257
- Zhu Y, Wang X, Hu M, Yang T, Xu H, Kang X, Chen X, Jiang L, Gao R, Wang J: Targeting Aβ and p-Tau Clearance in Methamphetamine-Induced Alzheimer's Disease-Like Pathology: Roles of Syntaxin 17 in Autophagic Degradation in Primary Hippocampal Neurons. Oxid Med Cell Longev 2022;2022:3344569.https://doi.org/xxxx10.1155/2022/3344569
- Ding J, Huang J, Xia B, Hu S, Fan H, Dai J, Li Z, Wang J, Le C, Qiu P, Wang Y: Transfer of α-synuclein from neurons to oligodendrocytes triggers myelin sheath destruction in methamphetamine administration mice. Toxicol Lett 2021;352:34-45.https://doi.org/xxxx10.1016/j.toxlet.2021.09.005
- Xu H, Zhu Y, Chen X, Yang T, Wang X, Song X, Xie X, Hu M, Jiang L, Cheng J, Gao R, Wang J: Mystery of methamphetamine-induced autophagosome accumulation in hippocampal neurons: loss of syntaxin 17 in defects of dynein-dynactin driving and autophagosome-late endosome/lysosome fusion. Arch Toxicol 2021;95:3263-3284.https://doi.org/xxxx10.1007/s00204-021-03131-y
- Yang G, Li J, Leung CK, Shen B, Wang C, Xu Y, Lin S, Zhang S, Tan Y, Zhang H, Zeng X, Hong S, Li L: Methamphetamine and HIV-1 Tat proteins synergistically induce microglial autophagy via activation of the Nrf2/NQO1/HO-1 signal pathway. Neuropharmacology 2022;220:109256.https://doi.org/xxxx10.1016/j.neuropharm.2022.109256
- Zare N, Maghsoudi N, Mirbehbahani SH, Foolad F, Khakpour S, Mansouri Z, Khodagholi F, Ghorbani Yekta B: Prenatal Methamphetamine Hydrochloride Exposure Leads to Signal Transduction Alteration and Cell Death in the Prefrontal Cortex and Amygdala of Male and Female Rats' Offspring. J Mol Neurosci 2022;72:2233-2241.https://doi.org/xxxx10.1007/s12031-022-02062-2
- Chen LJ, Zhi X, Zhang KK, Wang LB, Li JH, Liu JL, Xu LL, Yoshida JS, Xie XL, Wang Q: Escalating dose-multiple binge methamphetamine treatment elicits neurotoxicity, altering gut microbiota and fecal metabolites in mice. Food Chem Toxicol 2021;148:111946.https://doi.org/xxxx10.1016/j.fct.2020.111946
- Komlao P, Kraiwattanapirom N, Promyo K, Hein ZM, Chetsawang B: Melatonin enhances the restoration of neurological impairments and cognitive deficits during drug withdrawal in methamphetamine-induced toxicity and endoplasmic reticulum stress in rats. Neurotoxicology 2023;99:305-312.https://doi.org/xxxx10.1016/j.neuro.2023.11.006
- Mehrzadi S, Pourhanifeh MH, Mirzaei A, Moradian F, Hosseinzadeh A: An updated review of mechanistic potentials of melatonin against cancer: pivotal roles in angiogenesis, apoptosis, autophagy, endoplasmic reticulum stress and oxidative stress. Cancer Cell Int 2021;21:188.https://doi.org/xxxx10.1186/s12935-021-01892-1
- Hossain MF, Wang N, Chen R, Li S, Roy J, Uddin MG, Li Z, Lim LW, Song YQ: Exploring the multifunctional role of melatonin in regulating autophagy and sleep to mitigate Alzheimer's disease neuropathology. Ageing Res Rev 2021;67:101304.https://doi.org/xxxx10.1016/j.arr.2021.101304
- Demirkesen Ş, İriağaç Y, Şeber ES, Aral C: Melatonin enhances everolimus efficacy in breast cancer by suppressing mTOR pathway activation and promoting apoptosis and mitochondrial function. BMC Pharmacol Toxicol 2025;26:100.https://doi.org/xxxx10.1186/s40360-025-00907-1
- Luo F, Sandhu AF, Rungratanawanich W, Williams GE, Akbar M, Zhou S, Song BJ, Wang X: Melatonin and Autophagy in Aging-Related Neurodegenerative Diseases. Int J Mol Sci 2020;21https://doi.org/xxxx10.3390/ijms21197174
- Ding J, Shen L, Ye Y, Hu S, Ren Z, Liu T, Dai J, Li Z, Wang J, Luo Y, Zhang Q, Zhang X, Qi X, Huang J: Inflammasome Inhibition Prevents Motor Deficit and Cerebellar Degeneration Induced by Chronic Methamphetamine Administration. Front Mol Neurosci 2022;15:861340.https://doi.org/xxxx10.3389/fnmol.2022.861340
- Dutta D, Liu J, Xu E, Xiong H: Methamphetamine Enhancement of HIV-1 gp120-Mediated NLRP3 Inflammasome Activation and Resultant Proinflammatory Responses in Rat Microglial Cultures. Int J Mol Sci 2024;25https://doi.org/xxxx10.21203/rs.3.rs-3707515/v1
- Shen Y, Gong X, Qian L, Ruan Y, Lin S, Yu Z, Si Z, Wei W, Liu Y: Inhibition of GSDMD-dependent pyroptosis decreased methamphetamine self-administration in rats. Brain Behav Immun 2024;120:167-180.https://doi.org/xxxx10.1016/j.bbi.2024.05.040
- Fan R, Shen Y, Li X, Luo H, Zhang P, Liu Y, Si Z, Zhou W, Liu Y: The effect of the NLRP1 inflammasome on methamphetamine-induced cognitive impairment in rats. Drug Alcohol Depend 2022;237:109537.https://doi.org/xxxx10.1016/j.drugalcdep.2022.109537
- Zhang KK, Liu JL, Chen LJ, Li JH, Yang JZ, Xu LL, Chen YK, Zhang QY, Li XW, Liu Y, Zhao D, Xie XL, Wang Q: Gut microbiota mediates methamphetamine-induced hepatic inflammation via the impairment of bile acid homeostasis. Food Chem Toxicol 2022;166:113208.https://doi.org/xxxx10.1016/j.fct.2022.113208
- Wang LB, Xu LL, Chen LJ, Zhang KK, Zhang QY, Chen YK, Li JH, Liu JL, Wang Q, Xie XL: Methamphetamine induces intestinal injury by altering gut microbiota and promoting inflammation in mice. Toxicol Appl Pharmacol 2022;443:116011.https://doi.org/xxxx10.1016/j.taap.2022.116011
- Wang ZY, Guo LK, Han X, Song R, Dong GM, Ma CM, Wu N, Li J: Naltrexone attenuates methamphetamine-induced behavioral sensitization and conditioned place preference in mice. Behav Brain Res 2021;399:112971.https://doi.org/xxxx10.1016/j.bbr.2020.112971
- Li H, Peng Y, Lin C, Zhang X, Zhang T, Wang Y, Li Y, Wu S, Wang H, Hutchinson MR, Watkins LR, Wang X: Nicotine and its metabolite cotinine target MD2 and inhibit TLR4 signaling. Innovation (Camb) 2021;2:100111.https://doi.org/xxxx10.1016/j.xinn.2021.100111
- Wang LB, Chen LJ, Wang Q, Xie XL: Silencing the Tlr4 Gene Alleviates Methamphetamine-Induced Hepatotoxicity by Inhibiting Lipopolysaccharide-Mediated Inflammation in Mice. Int J Mol Sci 2022;23https://doi.org/xxxx10.3390/ijms23126810
- Ashrafizadeh M, Najafi M, Kavyiani N, Mohammadinejad R, Farkhondeh T, Samarghandian S: Anti-Inflammatory Activity of Melatonin: a Focus on the Role of NLRP3 Inflammasome. Inflammation 2021;44:1207-1222.https://doi.org/xxxx10.1007/s10753-021-01428-9
- Rashidi SK, Ansari Dezfouli M, Khodagholi F, Dadashpour M, Shabani AA: Melatonin Attenuates MethamphetamineNeurotoxicity through inhibition of NLRP3 andpyroptosis pathway. Physiology and Pharmacology 2024;28:430-439.https://doi.org/xxxx10.61186/phypha.28.4.430
- Chen H, Deng J, Gao H, Song Y, Zhang Y, Sun J, Zhai J: Involvement of the SIRT1-NLRP3 pathway in the inflammatory response. Cell Commun Signal 2023;21:185.https://doi.org/xxxx10.1186/s12964-023-01177-2
- Che H, Li H, Li Y, Wang YQ, Yang ZY, Wang RL, Wang LH: Melatonin exerts neuroprotective effects by inhibiting neuronal pyroptosis and autophagy in STZ-induced diabetic mice. Faseb j 2020;34:14042-14054.https://doi.org/xxxx10.1096/fj.202001328R
- Zhao Z, Wang X, Zhang R, Ma B, Niu S, Di X, Ni L, Liu C: Melatonin attenuates smoking-induced atherosclerosis by activating the Nrf2 pathway via NLRP3 inflammasomes in endothelial cells. Aging (Albany NY) 2021;13:11363-11380.https://doi.org/xxxx10.18632/aging.202829
- Zhang Y, Huang Y, Guo L, Zhang Y, Zhao M, Xue F, Tan C, Huang J, Chen D: Melatonin Alleviates Pyroptosis of Retinal Neurons Following Acute Intraocular Hypertension. CNS Neurol Disord Drug Targets 2021;20:285-297.https://doi.org/xxxx10.2174/1871527319666201012125149
- Ye D, Xu Y, Shi Y, Fan M, Lu P, Bai X, Feng Y, Hu C, Cui K, Tang X, Liao J, Huang W, Xu F, Liang X, Huang J: Anti-PANoptosis is involved in neuroprotective effects of melatonin in acute ocular hypertension model. J Pineal Res 2022;73:e12828.https://doi.org/xxxx10.1111/jpi.12828
- El-Sisi AEE, Sokar SS, Shebl AM, Mohamed DZ, Abu-Risha SE: Octreotide and melatonin alleviate inflammasome-induced pyroptosis through inhibition of TLR4-NF-κB-NLRP3 pathway in hepatic ischemia/reperfusion injury. Toxicol Appl Pharmacol 2021;410:115340.https://doi.org/xxxx10.1016/j.taap.2020.115340
- Liu Q, Sun Y, Zhu Y, Qiao S, Cai J, Zhang Z: Melatonin relieves liver fibrosis induced by Txnrd3 knockdown and nickel exposure via IRE1/NF-kB/NLRP3 and PERK/TGF-β1 axis activation. Life Sci 2022;301:120622.https://doi.org/xxxx10.1016/j.lfs.2022.120622
- Su ZD, Wei XB, Fu YB, Xu J, Wang ZH, Wang Y, Cao JF, Huang JL, Yu DQ: Melatonin alleviates lipopolysaccharide-induced myocardial injury by inhibiting inflammation and pyroptosis in cardiomyocytes. Ann Transl Med 2021;9:413.https://doi.org/xxxx10.21037/atm-20-8196
- Wen L, Wang M, Luo P, Meng X, Zhao M: Melatonin Exerts Cardioprotective Effects by Inhibiting NLRP3 Inflammasome-Induced Pyroptosis in Mice following Myocardial Infarction. Oxid Med Cell Longev 2021;2021:5387799.https://doi.org/xxxx10.1155/2021/5387799
- Lu J, Xu L, Zeng Z, Xue C, Li J, Chen X, Zhou P, Lin S, Liao Y, Du X, Yang R, Zheng S: Normothermic ex vivo Heart Perfusion Combined With Melatonin Enhances Myocardial Protection in Rat Donation After Circulatory Death Hearts via Inhibiting NLRP3 Inflammasome-Mediated Pyroptosis. Front Cell Dev Biol 2021;9:733183.https://doi.org/xxxx10.3389/fcell.2021.733183
- Wang YH, Gao X, Tang YR, Yu Y, Sun MJ, Chen FQ, Li Y: The Role of NF-κB/NLRP3 Inflammasome Signaling Pathway in Attenuating Pyroptosis by Melatonin Upon Spinal Nerve Ligation Models. Neurochem Res 2022;47:335-346.https://doi.org/xxxx10.1007/s11064-021-03450-7
- Ding F, Zhang L, Wu X, Liu Y, Wan Y, Hu J, Zhang X, Wu Q: Melatonin ameliorates renal dysfunction in glyphosate- and hard water-treated mice. Ecotoxicol Environ Saf 2022;241:113803.https://doi.org/xxxx10.1016/j.ecoenv.2022.113803
- Zhang C, Qiu Y, Yuan F: The long non-coding RNA maternally expressed 3-micorRNA-15a-5p axis is modulated by melatonin and prevents nucleus pulposus cell inflammation and apoptosis. Basic Clin Pharmacol Toxicol 2023;133:603-619.https://doi.org/xxxx10.1111/bcpt.13939
- Yu Z, Jiang N, Su W, Zhuo Y: Necroptosis: A Novel Pathway in Neuroinflammation. Front Pharmacol 2021;12:701564.https://doi.org/xxxx10.3389/fphar.2021.701564
- Kumar S, Budhathoki S, Oliveira CB, Kahle AD, Calhan OY, Lukens JR, Deppmann CD: Role of the caspase-8/RIPK3 axis in Alzheimer's disease pathogenesis and Aβ-induced NLRP3 inflammasome activation. JCI Insight 2023;8https://doi.org/xxxx10.1172/jci.insight.157433
- Li S, Qu L, Wang X, Kong L: Novel insights into RIPK1 as a promising target for future Alzheimer's disease treatment. Pharmacol Ther 2022;231:107979.https://doi.org/xxxx10.1016/j.pharmthera.2021.107979
- Oliveira SR, Dionísio PA, Gaspar MM, Correia Guedes L, Coelho M, Rosa MM, Ferreira JJ, Amaral JD, Rodrigues CMP: miR-335 Targets LRRK2 and Mitigates Inflammation in Parkinson's Disease. Front Cell Dev Biol 2021;9:661461.https://doi.org/xxxx10.3389/fcell.2021.661461
- Wang ZY, Zhen ZD, Fan DY, Wang PG, An J: Transcriptomic Analysis Suggests the M1 Polarization and Launch of Diverse Programmed Cell Death Pathways in Japanese Encephalitis Virus-Infected Macrophages. Viruses 2020;12https://doi.org/xxxx10.3390/v12030356
- Jeffries AM, Suptela AJ, Marriott I: Z-DNA binding protein 1 mediates necroptotic and apoptotic cell death pathways in murine astrocytes following herpes simplex virus-1 infection. J Neuroinflammation 2022;19:109.https://doi.org/xxxx10.1186/s12974-022-02469-z
- Yawoot N, Chumboatong W, Sengking J, Tocharus C, Tocharus J: Chronic high-fat diet consumption exacerbates pyroptosis- and necroptosis-mediated HMGB1 signaling in the brain after ischemia and reperfusion injury. J Physiol Biochem 2022;78:833-844.https://doi.org/xxxx10.1007/s13105-022-00906-4
- Hu Y, Lei H, Zhang S, Ma J, Kang S, Wan L, Li F, Zhang F, Sun T, Zhang C, Li W: Panax notoginseng Saponins Protect Brain Microvascular Endothelial Cells against Oxygen-Glucose Deprivation/Resupply-Induced Necroptosis via Suppression of RIP1-RIP3-MLKL Signaling Pathway. Neurochem Res 2022;47:3261-3271.https://doi.org/xxxx10.1007/s11064-022-03675-0
- Liu M, Li H, Yang R, Ji D, Xia X: GSK872 and necrostatin-1 protect retinal ganglion cells against necroptosis through inhibition of RIP1/RIP3/MLKL pathway in glutamate-induced retinal excitotoxic model of glaucoma. J Neuroinflammation 2022;19:262.https://doi.org/xxxx10.1186/s12974-022-02626-4
- Xu W, Yang T, Lou X, Chen J, Wang X, Hu M, An D, Gao R, Wang J, Chen X: Role of the Peli1-RIPK1 Signaling Axis in Methamphetamine-Induced Neuroinflammation. ACS Chem Neurosci 2023;14:864-874.https://doi.org/xxxx10.1021/acschemneuro.2c00623
- Guo LM, Wang Z, Li SP, Wang M, Yan WT, Liu FX, Wang CD, Zhang XD, Chen D, Yan J, Xiong K: RIP3/MLKL-mediated neuronal necroptosis induced by methamphetamine at 39°C. Neural Regen Res 2020;15:865-874.https://doi.org/xxxx10.4103/1673-5374.268902
- Zhou P, Xuan J, Xu W, An D, Meng S, Zhang H, Hu M, Hui W, Wang Y, Cheng J, Xiong J, Wang J, Chen X: Methamphetamine Exposure Induces Neuronal Programmed Necrosis by Permeabilizing Mitochondria via the RIPK1-RIPK3-MLKL Axis. Toxics 2025;13https://doi.org/xxxx10.3390/toxics13090736
- Liao LS, Lu S, Yan WT, Wang SC, Guo LM, Yang YD, Huang K, Hu XM, Zhang Q, Yan J, Xiong K: The Role of HSP90α in Methamphetamine/Hyperthermia-Induced Necroptosis in Rat Striatal Neurons. Front Pharmacol 2021;12:716394.https://doi.org/xxxx10.3389/fphar.2021.716394
- Mao K, Luo P, Geng W, Xu J, Liao Y, Zhong H, Ma P, Tan Q, Xia H, Duan L, Song S, Long D, Liu Y, Yang T, Wu Y, Jin Y: An Integrative Transcriptomic and Metabolomic Study Revealed That Melatonin Plays a Protective Role in Chronic Lung Inflammation by Reducing Necroptosis. Front Immunol 2021;12:668002.https://doi.org/xxxx10.3389/fimmu.2021.668002
- Sheibani M, Hosseinzadeh A, Fatemi I, Mehrzadi S: Melatonin and necroptosis: therapeutic aspects based on cellular mechanisms. Mol Biol Rep 2025;52:606.https://doi.org/xxxx10.1007/s11033-025-10713-x
- Qiu M, Zhang W, Dai J, Sun W, Lai M, Tang S, Xu E, Ning Y, Zhan L: A20 negatively regulates necroptosis-induced microglia/macrophages polarization and mediates cerebral ischemic tolerance via inhibiting the ubiquitination of RIPCell Death Dis 2024;15:904.https://doi.org/xxxx10.1038/s41419-024-07293-2
- Hassanein EHM, Ibrahim IM, Abd El-Maksoud MS, Abd El-Aziz MK, Abd-Alhameed EK, Althagafy HS: Targeting necroptosis in fibrosis. Mol Biol Rep 2023;50:10471-10484.https://doi.org/xxxx10.1007/s11033-023-08857-9
- Bhardwaj M, Upmanyu K, Upadhyay S: PANoptosis as a drug discovery framework: integrating cell death architecture with clinical translation. Genes Immun 2026https://doi.org/xxxx10.1038/s41435-026-00388-0
- Nguyen LN, Kanneganti TD: PANoptosis in Viral Infection: The Missing Puzzle Piece in the Cell Death Field. J Mol Biol 2022;434:167249.https://doi.org/xxxx10.1016/j.jmb.2021.167249
- Paidlewar M, Kumari S, Dhapola R, Sharma P, Vellingiri B, Medhi B, HariKrishnaReddy D: PANoptosis in Alzheimer's disease: The expanding landscape of programmed cell death mechanisms and therapeutic interventions. Free Radic Biol Med 2026https://doi.org/xxxx10.1016/j.freeradbiomed.2026.03.053
- Paranga TG, Mitu I, Pavel-Tanasa M, Rosu MF, Miftode IL, Constantinescu D, Obreja M, Plesca CE, Miftode E: Cytokine Storm in COVID-19: Exploring IL-6 Signaling and Cytokine-Microbiome Interactions as Emerging Therapeutic Approaches. Int J Mol Sci 2024;25https://doi.org/xxxx10.3390/ijms252111411
- Yuan L, Wang B, Huang YW: Regulation of Apoptosis and PANoptosis by Coronavirus: An Overview. J Med Virol 2025;97:e70672.https://doi.org/xxxx10.1002/jmv.70672
- Yang B, Hu A, Wang T, Chen X, Ma C, Yang X, Deng K: SARS-CoV-2 infection induces ZBP1-dependent PANoptosis in bystander cells. Proc Natl Acad Sci U S A 2025;122:e2500208122.https://doi.org/xxxx10.1073/pnas.2500208122
- Eltaib L, Alanazi MN, Khan Y, Hanbashi A, Khan G, Rikabi HA, Maqbool M, Hussain MS: PANoptosis in Cytokine Release Syndrome: Bridging the Gap between Inflammation and Cell Death. Curr Pharm Des 2025https://doi.org/xxxx10.2174/0113816128404910250912133524
- Ma C, Zhou X, Pan S, Liu L: AIM2 mediated neuron PANoptosis plays an important role in diabetes cognitive dysfunction. Behav Brain Res 2025;491:115651.https://doi.org/xxxx10.1016/j.bbr.2025.115651
- Zhang W, Zhu C, Liao Y, Zhou M, Xu W, Zou Z: Caspase-8 in inflammatory diseases: a potential therapeutic target. Cell Mol Biol Lett 2024;29:130.https://doi.org/xxxx10.1186/s11658-024-00646-x
- Cui Y, Hu Z, Wang L, Zhu B, Deng L, Zhang H, Wang X: DL-3-n-Butylphthalide Ameliorates Post-stroke Emotional Disorders by Suppressing Neuroinflammation and PANoptosis. Neurochem Res 2024;49:2215-2227.https://doi.org/xxxx10.1007/s11064-024-04171-3
- Rajesh Y, Kanneganti TD: Innate Immune Cell Death in Neuroinflammation and Alzheimer's Disease. Cells 2022;11https://doi.org/xxxx10.3390/cells11121885
- Pandeya A, Kanneganti TD: Therapeutic potential of PANoptosis: innate sensors, inflammasomes, and RIPKs in PANoptosomes. Trends Mol Med 2024;30:74-88.https://doi.org/xxxx10.1016/j.molmed.2023.10.001
- He F, Liu C, Yu Y, Wang Q, Li L, Chao X, Chen L, Yu C: Melatonin Suppresses NLRP3 Inflammasome Activation via SIRT1-Mediated ETS1 Deacetylation to Attenuate LPS-Induced Pyroptosis in Alveolar Epithelial Cells. J Inflamm Res 2026;19:581957.https://doi.org/xxxx10.2147/JIR.S581957
- Hu S, Huang X, Huang J, Qian Y, Tian Y, Xiao Y, Qi X, Zhou X, Yang Z, Chen Z: Iron chelation prevents nigrostriatal neurodegeneration in a chronic methamphetamine mice model. Neurotoxicology 2023;99:24-33.https://doi.org/xxxx10.1016/j.neuro.2023.09.006
- Zhou J, Xu Z, Peng DB, Li X, Chang S, Duan K, Jiang Y, Gu C, Peng X, Xie WB: Macrophage-Derived LCN2 Promotes Methamphetamine-Induced Pulmonary Hypertension. Hypertension 2025;82:1355-1367.https://doi.org/xxxx10.1161/HYPERTENSIONAHA.124.24548
- Lin S, Cheng H, Yang G, Wang C, Leung CK, Zhang S, Tan Y, Zhang H, Wang H, Miao L, Li Y, Huang Y, Li J, Zhang R, Zeng X: NRF2 Antagonizes HIV-1 Tat and Methamphetamine-Induced BV2 Cell Ferroptosis by Regulating SLC7ANeurotox Res 2023;41:398-407.https://doi.org/xxxx10.1007/s12640-023-00645-4
- Lin S, Cheng H, Yang G, Wang C, Leung CK, Zhang S, Tan Y, Zhang H, Wang H, Miao L, Li Y, Huang Y, Li J, Zhang R, Zeng X: NRF2 Antagonizes HIV-1 Tat and Methamphetamine-Induced BV2 Cell Ferroptosis by Regulating SLC7ANeurotox Res 2023https://doi.org/xxxx10.1007/s12640-023-00645-4
- Guo C, Chen L, Wang Y: Substance abuse and neurodegenerative diseases: focus on ferroptosis. Arch Toxicol 2023https://doi.org/xxxx10.1007/s00204-023-03505-4
- Wang X, Wang Z, Cao J, Dong Y, Chen Y: Melatonin Alleviates Acute Sleep Deprivation-Induced Memory Loss in Mice by Suppressing Hippocampal Ferroptosis. Front Pharmacol 2021;12:708645.https://doi.org/xxxx10.3389/fphar.2021.708645
- Gou Z, Su X, Hu X, Zhou Y, Huang L, Fan Y, Li J, Lu L: Melatonin improves hypoxic-ischemic brain damage through the Akt/Nrf2/Gpx4 signaling pathway. Brain Res Bull 2020;163:40-48.https://doi.org/xxxx10.1016/j.brainresbull.2020.07.011
- Ma H, Wang X, Zhang W, Li H, Zhao W, Sun J, Yang M: Melatonin Suppresses Ferroptosis Induced by High Glucose via Activation of the Nrf2/HO-1 Signaling Pathway in Type 2 Diabetic Osteoporosis. Oxid Med Cell Longev 2020;2020:9067610.https://doi.org/xxxx10.1155/2020/9067610
- Huang YB, Jiang L, Liu XQ, Wang X, Gao L, Zeng HX, Zhu W, Hu XR, Wu YG: Melatonin Alleviates Acute Kidney Injury by Inhibiting NRF2/Slc7a11 Axis-Mediated Ferroptosis. Oxid Med Cell Longev 2022;2022:4776243.https://doi.org/xxxx10.1155/2022/4776243
- Li M, Yang N, Hao L, Zhou W, Li L, Liu L, Yang F, Xu L, Yao G, Zhu C, Xu W, Fang S: Melatonin Inhibits the Ferroptosis Pathway in Rat Bone Marrow Mesenchymal Stem Cells by Activating the PI3K/AKT/mTOR Signaling Axis to Attenuate Steroid-Induced Osteoporosis. Oxid Med Cell Longev 2022;2022:8223737.https://doi.org/xxxx10.1155/2022/8223737
- Guohua F, Tieyuan Z, Xinping M, Juan X: Melatonin protects against PM2.5-induced lung injury by inhibiting ferroptosis of lung epithelial cells in a Nrf2-dependent manner. Ecotoxicol Environ Saf 2021;223:112588.https://doi.org/xxxx10.1016/j.ecoenv.2021.112588
- Naidu SAG, Clemens RA, Naidu AS: SARS-CoV-2 Infection Dysregulates Host Iron (Fe)-Redox Homeostasis (Fe-R-H): Role of Fe-Redox Regulators, Ferroptosis Inhibitors, Anticoagulants, and Iron-Chelators in COVID-19 Control. J Diet Suppl 2022:1-60.https://doi.org/xxxx10.1080/19390211.2022.2075072
- Qiu W, An S, Wang T, Li J, Yu B, Zeng Z, Chen Z, Lin B, Lin X, Gao Y: Melatonin suppresses ferroptosis via activation of the Nrf2/HO-1 signaling pathway in the mouse model of sepsis-induced acute kidney injury. Int Immunopharmacol 2022;112:109162.https://doi.org/xxxx10.1016/j.intimp.2022.109162
- Yang J, Tang Q, Zeng Y: Melatonin: Potential avenue for treating iron overload disorders. Ageing Res Rev 2022;81:101717.https://doi.org/xxxx10.1016/j.arr.2022.101717
- Mi Y, Wei C, Sun L, Liu H, Zhang J, Luo J, Yu X, He J, Ge H, Liu P: Melatonin inhibits ferroptosis and delays age-related cataract by regulating SIRT6/p-Nrf2/GPX4 and SIRT6/NCOA4/FTH1 pathways. Biomed Pharmacother 2023;157:114048.https://doi.org/xxxx10.1016/j.biopha.2022.114048
- Cheng Y, Gao Y, Li J, Rui T, Li Q, Chen H, Jia B, Song Y, Gu Z, Wang T, Gao C, Wang Y, Wang Z, Wang F, Tao L, Luo C: TrkB agonist N-acetyl serotonin promotes functional recovery after traumatic brain injury by suppressing ferroptosis via the PI3K/Akt/Nrf2/Ferritin H pathway. Free Radic Biol Med 2023;194:184-198.https://doi.org/xxxx10.1016/j.freeradbiomed.2022.12.002
- Kozuka K, Egami R, Gao TX, Ohsawa M, Tomita J, Kume K: Aripiprazole advances delayed sleep-wake rhythm in methamphetamine-treated mice. Chronobiol Int 2026;43:172-182.https://doi.org/xxxx10.1080/07420528.2025.2591925
- Liu Y, Wang Y, Liu J, Kang R, Tang D: The circadian clock protects against ferroptosis-induced sterile inflammation. Biochem Biophys Res Commun 2020;525:620-625.https://doi.org/xxxx10.1016/j.bbrc.2020.02.142
- Chen X, Yu C, Kang R, Kroemer G, Tang D: Cellular degradation systems in ferroptosis. Cell Death Differ 2021;28:1135-1148.https://doi.org/xxxx10.1038/s41418-020-00728-1
- Liu J, Yang M, Kang R, Klionsky DJ, Tang D: Autophagic degradation of the circadian clock regulator promotes ferroptosis. Autophagy 2019;15:2033-2035.https://doi.org/xxxx10.1080/15548627.2019.1659623
- Chen Y, Guan S, Liu M, Lang L, Peng H, Lu J: 1, 3-Dichloro-2-propanol Induced Renal Cell Ferroptosis via the Circadian Clock Protein BMAL1 Targeting GPXJ Agric Food Chem 2024;72:26859-26873.https://doi.org/xxxx10.1021/acs.jafc.4c05676
- Xu S, Tang Q, Du H, Xie J, He R, Wang R, Sun Q: Mechanism of Circadian Regulation in Ferroptosis of the BMAL1/NRF2 Pathway in Renal Ischemia-Reperfusion. Biomedicines 2025;13https://doi.org/xxxx10.3390/biomedicines13061375
- Lin H, Ji F, Lin KQ, Zhu YT, Yang W, Zhang LH, Zhao JG, Pei YH: LPS-aggravated Ferroptosis via Disrupting Circadian Rhythm by Bmal1/AKT/p53 in Sepsis-Induced Myocardial Injury. Inflammation 2023;46:1133-1143.https://doi.org/xxxx10.1007/s10753-023-01804-7
- Zhao Y, Zhang R, Wang Z, Chen Z, Wang G, Guan S, Lu J: Melatonin Prevents against Ethanol-Induced Liver Injury by Mitigating Ferroptosis via Targeting Brain and Muscle ARNT-like 1 in Mice Liver and HepG2 Cells. J Agric Food Chem 2022;70:12953-12967.https://doi.org/xxxx10.1021/acs.jafc.2c04337
- Guo L, Zhang T, Wang F, Chen X, Xu H, Zhou C, Chen M, Yu F, Wang S, Yang D, Wu B: Targeted inhibition of Rev-erb-α/β limits ferroptosis to ameliorate folic acid-induced acute kidney injury. Br J Pharmacol 2021;178:328-345.https://doi.org/xxxx10.1111/bph.15283
- Huang Q, Tian L, Zhao X, Lei S, Zhao B, Qiu Z, Xia ZY: Rev-erbs agonist SR9009 alleviates ischemia-reperfusion injury by heightening endogenous cardioprotection at onset of type-2 diabetes in rats: Down-regulating ferritinophagy/ferroptosis signaling. Biomed Pharmacother 2022;154:113595.https://doi.org/xxxx10.1016/j.biopha.2022.113595
- Yang Y, Tang H, Zheng J, Yang K: The PER1/HIF-1alpha negative feedback loop promotes ferroptosis and inhibits tumor progression in oral squamous cell carcinoma. Transl Oncol 2022;18:101360.https://doi.org/xxxx10.1016/j.tranon.2022.101360
- Shu YY, Gao WK, Chu HK, Yang L, Pan XL, Ye J: Attenuation by Time-Restricted Feeding of High-Fat and High-Fructose Diet-Induced NASH in Mice Is Related to Per2 and Ferroptosis. Oxid Med Cell Longev 2022;2022:8063897.https://doi.org/xxxx10.1155/2022/8063897
- Huang R, Pang Q, Shen D, Zheng L, Wang L, Jia B, Xu Y, Du J, Li Y, Dai F, Sun W, Shen Y, Gao C, Gu Z, Wang Z, Tao L, Wang T: BMAL1-mediated circadian-ferroptosis crosstalk drives neuronal vulnerability after TBI. Free Radic Biol Med 2026;243:548-563.https://doi.org/xxxx10.1016/j.freeradbiomed.2025.11.058
- Yehia A, Abulseoud OA: Melatonin: a ferroptosis inhibitor with potential therapeutic efficacy for the post-COVID-19 trajectory of accelerated brain aging and neurodegeneration. Mol Neurodegener 2024;19:36.https://doi.org/xxxx10.1186/s13024-024-00728-6
- Cruz-Sanabria F, Carmassi C, Bruno S, Bazzani A, Carli M, Scarselli M, Faraguna U: Melatonin as a Chronobiotic with Sleep-promoting Properties. Curr Neuropharmacol 2023;21:951-987.https://doi.org/xxxx10.2174/1570159X20666220217152617
- Anghel L, Baroiu L, Popazu CR, Pătraș D, Fotea S, Nechifor A, Ciubara A, Nechita L, Mușat CL, Stefanopol IA, Tatu AL, Ciubara AB: Benefits and adverse events of melatonin use in the elderly (Review). Exp Ther Med 2022;23:219.https://doi.org/xxxx10.3892/etm.2022.11142
- Givler D, Givler A, Luther PM, Wenger DM, Ahmadzadeh S, Shekoohi S, Edinoff AN, Dorius BK, Jean Baptiste C, Cornett EM, Kaye AM, Kaye AD: Chronic Administration of Melatonin: Physiological and Clinical Considerations. Neurol Int 2023;15:518-533.https://doi.org/xxxx10.3390/neurolint15010031